DPhil researcher Selena Milanovic works on mathematical models that aim to unpick the underlying mechanisms behind the causes of brain diseases like Alzheimer’s and dementia.
The brain is one of our most valuable organs. The control centre of the body, responsible for regulating our emotions, processing the world around us and storing our memories. Degenerative brain diseases such as Alzheimer’s and dementia are debilitating to those who have them and their families. Engineering DPhil researcher Selena Milanovic is working on mathematical models that could provide doctors with detailed insights that hold significant potential to identify and prevent brain degeneration.
As with most illnesses, the earlier you identify them, the better the chances are for treating the patient effectively. The scale at which degenerative diseases begin in the brain are so small that current imaging techniques are unable to capture them. However, an alternative way to access this information is through modelling. A model is a mathematical representation of a physical process, requiring inputs, e.g. measurements, which it uses to make a prediction.
“I am designing mathematical models that aim to show us what pictures hide and can help us explain the relationship between brain blood flow and nutrient consumption. My project is a segment of a greater objective of the Cerebral Haemodynamics Group (CHG) led by Professor Stephen Payne, aiming to describe neurological mechanisms using engineering tools. ”, says Milanovic.
A lack of blood flow (or ‘perfusion’) to certain areas of the brain can cause degenerative diseases. As Milanovic explains,
“The blood flow supplies the tissue with nutrients. Therefore, an inadequate blood perfusion of the tissue is associated with an insufficient nutrient supply, causing tissue death.”
The flow of blood to the brain is regulated based on three factors; oxygen supply, ‘pericyte cell’ activity and glucose consumption. Pericyte cells regulate blood flow to the brain as they contract and control the diameter of capillary vessels which transport blood. Milanovic has designed the first mathematical model of pericyte cell activity in the brain to show how they regulate blood flow given measurements of oxygen supply, “Ultimately, I aim to validate the model's predictions with human patient data.”.
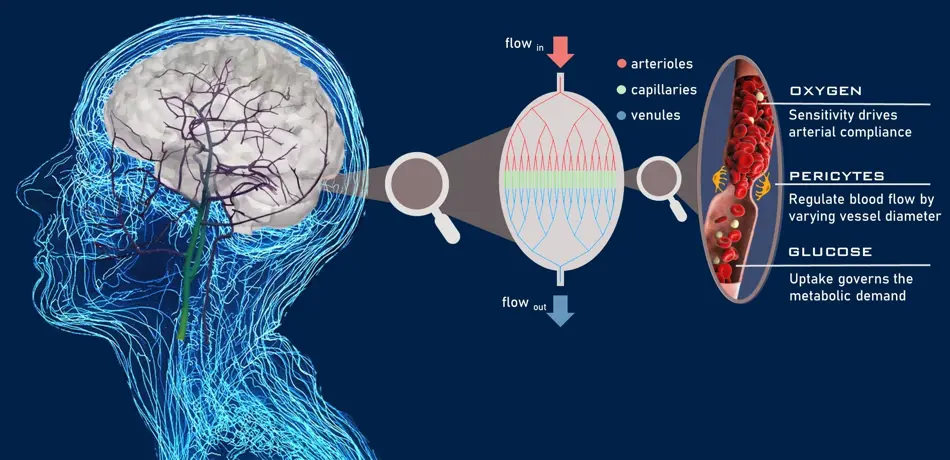
A diagram showing the roles of oxygen supply, pericyte cell activity and glucose consumption.
Previous models reproduce the physiological reactions of the brain using relationships between various aspects of the brain. Selena’s work focusses on constructing models of the underlying mechanisms to bridge with those of the physiological reactions that they cause.
When asked how she first got interested in Biomedical Engineering, Milanovic says : “Having a background in mechanical engineering, I had the opportunity to learn about the exceptional translatability of concepts within the engineering discipline. Indeed, during my undergraduate degree I learned that the mechanics of climbing plants can be used to design robotics applications. Similarly, the same principles used to describe the branching of rivers can be used to investigate the bifurcation of blood vessels. This cross-disciplinary research intrigued me greatly and I decided to pursue an MSc in Biomedical and Ecological sciences, during which I worked on designing artificial human lungs aimed to replace animal testing.”

Selena Milanovic is a DPhil researcher working with the Cerebral Haemodynamics Group at the Institute of Biomedical Engineering.
Milanovic’s DPhil work has led her to recently present her project in parliament as part of STEM for Britain, “In a technology-driven world, we often find a divide between scientific advance and policy implementation. STEM for Britain aims to bridge this gap, by offering a platform for communication between academic and government bodies. I aimed to use this opportunity to promote the potential of engineering techniques in a clinical context by highlighting their impact on UK healthcare. Moreover, I hope that my research and participation will incite more young girls to pursue a career in an environment that is still male dominated.”
Milanovic at STEM for Britain
In the future, it may even be possible to prevent serious advances of degenerative brain diseases. A goal of the research is to make it clinically applicable, such that it can be used directly to help patients while they are being treated. Current treatments that help with blood pressure and oxygenation levels can work in the short term, but there have been cases of bleeds and increased pressure within the skull that require surgical intervention. “Currently there is not a defined optimum perfusion pressure value, mainly because every brain is different and there is no ‘one size fits all’.”, explains Milanovic. The three metabolic regulators can be monitored, and so combined with the model will one day be able to give doctors pinpoint precision as to how each factor influences blood flow to ensure the best possible care for the patient.
Milanovic adds,
“Ultimately, what fuels my zest for engineering is the aim to improve people's quality of life. Particularly as a biomedical engineer, I strive to make an impact in the healthcare sector.”
Research Groups
How chemical treatments can change bamboo’s physical properties
Materials Engineering

